Our studies
Our strong confidence in the GASMAS technology and its application in the Neola® device for continuous lung monitoring has been built during the recent years and we have taken several steps to improve both the performance and the clinical acceptance. Read more about our work below.
If you want to know more about the technology, read our white paper.
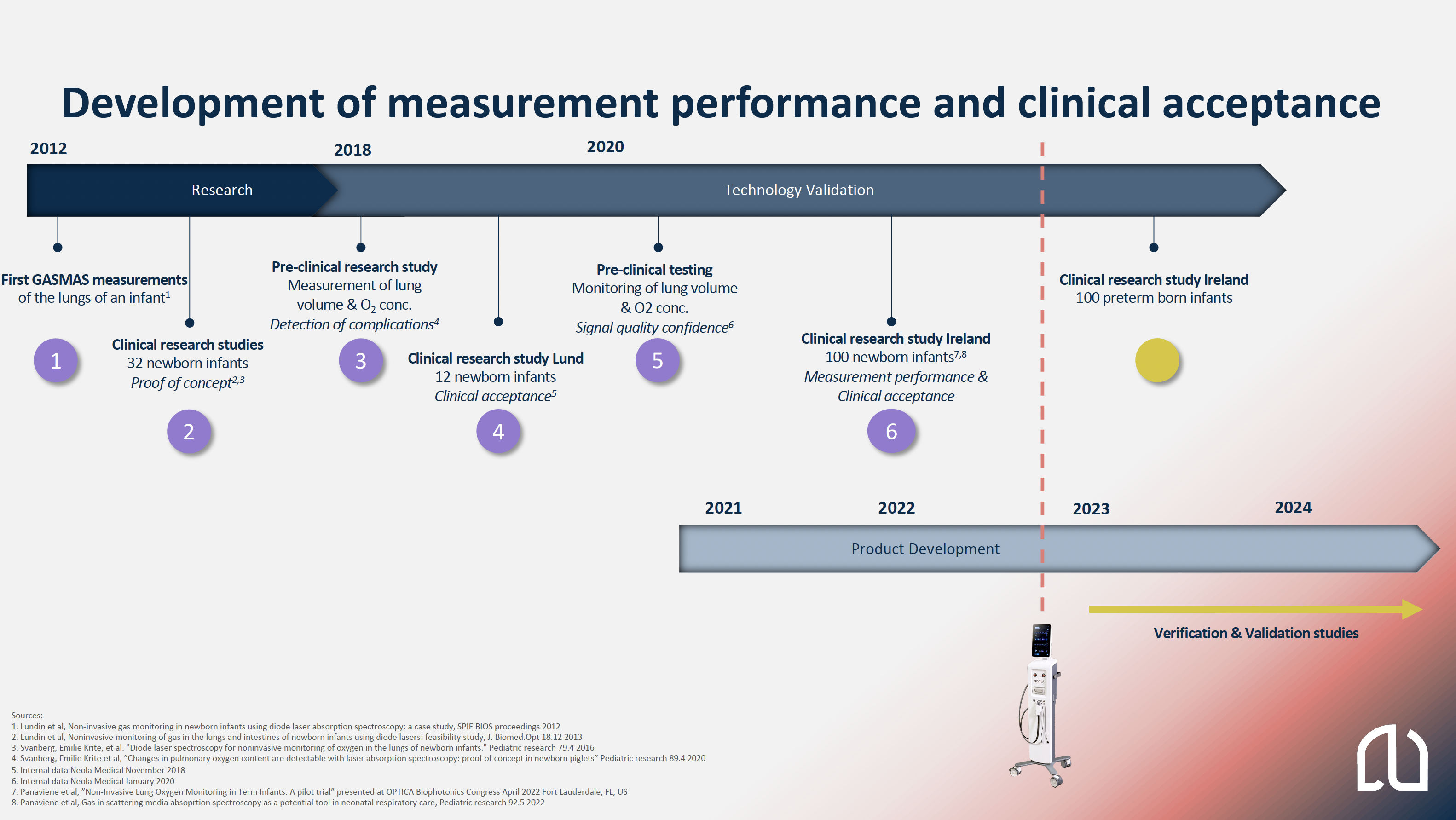
- In 2012, the first gas absorption measurement was performed on one infant followed by two more research studies in 2013. In total 32 infants were measured, and the results showed the feasibility of measuring gas in the the lungs of infants.
- In 2018, a pre-clinical study was performed, which showed the technology’s ability to measure oxygen gas concentrations in the lungs, follow changes in lung volume and to detect respiratory complications.
- A clinically adopted device was designed and in the end of 2018, a clinical study on 12 infants was performed in Lund, that showed clinical acceptance for the measurements in general and especially with the design of the probe, one of the most important perspectives to consider for this vulnerable patient group.
- The product development was initiated in late 2020, after improvements made to the electronics design in 2019.
- During 2021 and 2022, an investigator-initiated large clinical study on 100 newborn infants was performed at the INFANT center, University Hospital in Cork, Ireland. This study has further strengthened our confident in the performance of the measurements and the clinical acceptance and Neola®’s future role in Neonatal Intensive Care Unit. Read more about the study below.
- During 2023 Neola Medical is planning for further technical verification studies, usability studies and clinical validation studies.
-
Published articles
Previously published articles related to the technology of Neola Medical.
Non-Invasive Lung Oxygen Monitoring in Term Infants (NIOMI)
Tyndall National Institute at University College Cork in Ireland (UCC) aquired a research system with the Neola®-technology from Neola Medical AB (publ) in 2021 and started a large clinical study on newborn infants at the research facility at Cork University Maternity Hospital. The study was conducted in cooperation with The Irish Centre for Maternal and Child Health Research (INFANT) at UCC and included in total 100 newborn babies.
The aim of the study is to investigate the possibility to give real-time information about the lung function in newborn babies. The study examines the influence of the location of the Neola®-probes on different areas of the infants’ chests, which can provide valuable clinical information for using Neola® at neonatal intensive care units by doctors and nurses. The principal investigator of the study is Professor Eugene Dempsey, Horgan Chair in Neonatology, INFANT Centre, University College Cork (UCC), and he is supported by the BioPhotonics team at Tyndall.
Results and conclusions from the first 50 newborn infants were presented on the 25th of April 2022 by clinical researcher and neonatologist Jurate Panaviene at the OPTICA Biophotonic Congress: Biomedical Optics in Florida, the U.S. Read the press release about it here.
During the fall 2022, the clinical study was finalized after successful recruitment of 100 babies. The results and conclusions from the 100 newborn infants were presented at the jENS conference in Italy on the 23rd of September 2023, by clinical researcher and neonatologist Jurate Panaviene. Read the press release about it here.
Research work at Neola Medical
As part of the development process at Neola Medical, internal usability and performance studies is performed with nurses, neonatologists, dolls and with animal models to ensure performance, safety and usability.
For Investors
We are on a journey to revolutionize neonatal intensive care
Learn more about investing in Neola Medical